


Along the North Carolina coast, spring does not arrive all at once. It filters in through temperature gradients, longer light, and currents that shift almost imperceptibly until the water itself feels different. Animals respond before people do. Some move north. Some move inshore. Others arrive carrying a process already underway — reproduction unfolding quietly inside bodies designed to measure time in seasons rather than days.
This post explores shark reproduction in North Carolina, not as spectacle, but as a system of time, geography, and survival.
Shark reproduction is rarely visible. There are no surface displays, no spectacle to announce the moment. Instead, lineage advances through anatomical engineering and geographic choreography. The coastline becomes a corridor through which inheritance travels. What appears to be migration is often the hidden architecture of the next generation. Across shark species, reproductive strategies are tightly bound to life history pacing — longevity, growth rate, and investment per offspring — forming evolutionary solutions calibrated to risk and time (Cortés, 2000; Musick & Ellis, 2005).
Sharks do not share a single blueprint for reproduction. Some lay eggs encased in protective capsules that anchor to the seafloor. Others carry embryos internally and give birth to fully formed young. Between those extremes lies a spectrum of strategies — eggs retained inside the mother, embryos nourished in different ways, gestation stretched across seasons rather than weeks. The diversity is not incidental. It is the result of a lineage experimenting with how best to move the future through water: protect it externally, carry it internally, or invest in a few individuals built to survive from the first moment they enter open ocean (Carrier et al., 2012; Cortés, 2000).
Each winter, Atlantic spiny dogfish (Squalus acanthias) thin from our nearshore waters. Their absence is not disappearance but redistribution. Along the Northwest Atlantic coast the species occupies a broad range from Canada to the Carolinas, but this range is not a single undifferentiated mass. Seasonal movements reveal two general latitudinal tendencies — a northern contingent centered toward New England and Canadian waters, and a southern contingent extending toward North Carolina. In spring, portions of both groups converge in mid-Atlantic shelf waters, where overlapping migrations create temporary reproductive mixing before adults disperse again toward their habitual ranges (Carlson et al., 2014).
This convergence is not random drift. It is structured migration. Satellite tracking shows that spiny dogfish follow repeatable north–south circuits tied to temperature and habitat gradients rather than wandering opportunistically (Carlson et al., 2014). During these seasonal overlaps, sex and maturity stage influence where individuals position themselves within the shared corridor. Females and mature animals use space differently from juveniles, reflecting reproductive status and energetic demand (DeVries et al., 2025). The result is a coastline briefly braided by lineage: individuals from distant home waters exchanging genetic material before returning south or north to complete gestation.
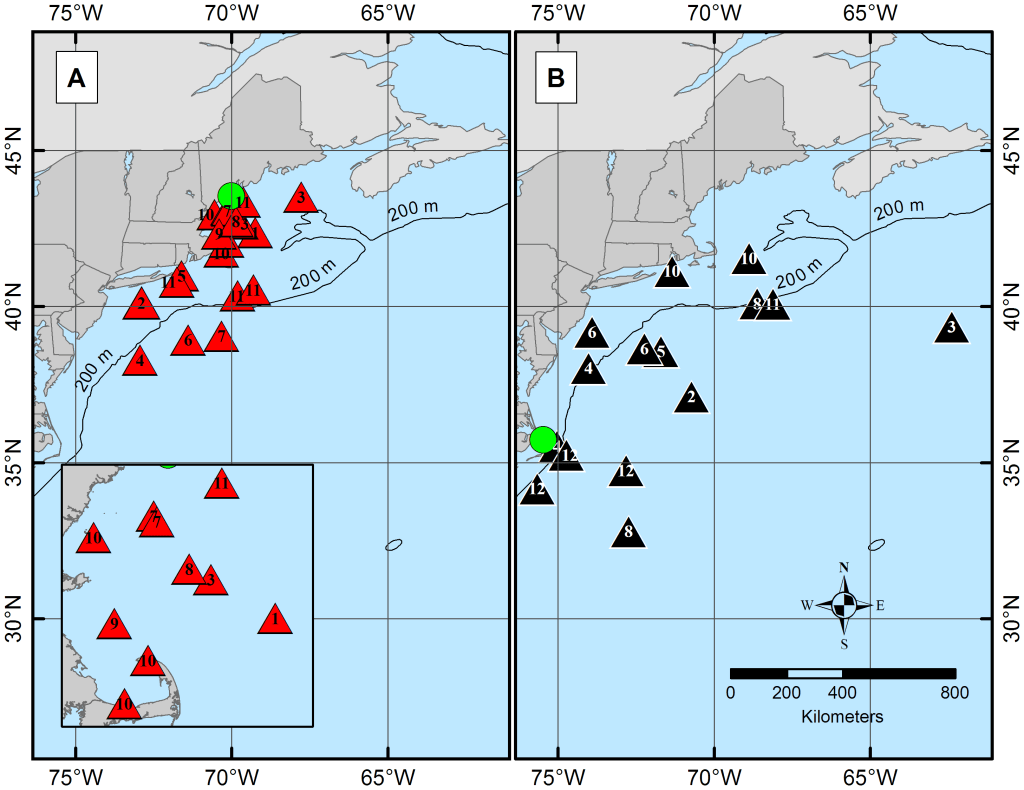
After fertilization, females carry embryos for nearly two years — among the longest gestation periods recorded in sharks (Hamlett, 2005). A single pregnancy produces relatively small litters, commonly averaging six to twelve pups, each representing a substantial maternal investment spread across seasons rather than weeks (Hamlett, 2005; Cortés, 2000). Birth does not occur in the same waters where mating took place. Instead, adults retreat toward their familiar temperature zones and feeding grounds, and the next generation enters the ocean already geographically sorted. Migration and reproduction form a loop rather than a point. Each cycle redistributes genes across the coast while preserving the regional rhythms that structure the population.
This extraordinary investment in time creates vulnerability. Sharks with slow growth, delayed maturity, and extended gestation replace themselves gradually, making populations sensitive to elevated fishing pressure (Cortés, 2000; Musick & Ellis, 2005). Removing a late-term female represents not a single loss, but the collapse of years of biological investment in a species evolved for endurance rather than speed.
Female sharks often carry scars along their flanks and fins — pale arcs and punctures that appear deliberate enough to invite explanation. These marks are frequently attributed to mating, and sometimes that interpretation is correct. During copulation, males grip females with their teeth to maintain position in moving water, producing patterned abrasions consistent with tooth spacing (Pratt & Carrier, 2005). But the body of a coastal predator is an archive of many encounters, not all of them reproductive.

Fishing gear produces different signatures: hooks damage the jaw, entanglement leaves constricting linear abrasions, and vessel strikes create irregular trauma. Healed injuries accumulate across a lifetime, recording survival rather than singular events. Marine biologists interpret these marks through context — season, species behavior, wound geometry — understanding that a scar is evidence, not confession (Pratt & Carrier, 2005). The ocean rarely supplies a single explanation.

Scars are only one layer of interpretation. Sharks also carry quieter markers of sex and maturity written into their form. Males develop elongated claspers — modified fins that trail beneath the body — visible even at a distance once the animal reaches reproductive age. In immature males these structures are short and flexible, almost decorative. With maturity they lengthen and calcify, projecting clearly behind the pelvic fins like paired shadows. A school viewed from a pier often reveals this difference in motion: some bodies carry that trailing geometry, others do not. Even without knowing species, an observer is watching a mixed population divided by sex and age.
Females, lacking claspers, present a cleaner silhouette. During pregnancy their bodies shift subtly. The abdomen rounds, not dramatically but enough to change how light moves across the flank. Experienced observers recognize gravid females less by size than by proportion — a redistribution of mass that suggests internal cargo rather than surface injury.
The clasper itself is an evolutionary innovation — a modification of pelvic fins that allows internal fertilization in a fluid environment where external fertilization would disperse gametes too widely to ensure success (Hamlett, 2005). It is a structural solution to a problem posed by water: how to keep lineage from dissolving into current.

These signals are quiet. They require patience. To read a shark in the water is to read a body moving through stages — juvenile, mature, gravid — each phase revealing that reproduction is not a single event but a condition carried across seasons. The distinction is anatomical literacy learned slowly, the way birdwatchers learn silhouettes or botanists learn leaf shape. Bodies announce their histories to those patient enough to look.
Maturity does not arrive uniformly across a population. In many coastal sharks, size is a better predictor of reproductive readiness than age. Warmer water accelerates metabolism and growth, allowing juveniles in southern nurseries to reach maturity sooner than their northern counterparts (Cortés, 2000; Musick & Ellis, 2005). Temperature becomes a developmental force. A difference of a few degrees can compress or extend the timeline by years, shaping when an individual enters the reproductive pool.
Juveniles and adults often sort themselves accordingly. Young sharks cluster in shallower, warmer margins where rapid growth offsets vulnerability. Larger, mature individuals occupy deeper or more exposed water, their size granting a margin of safety (Heupel et al., 2007). When mixed schools appear near piers, the variation in body shape reflects overlapping life stages sharing temporary habitat. What looks like a single group is often a layered demographic — future breeders moving alongside current ones.
During mating seasons, additional cues surface. Mature males display fully calcified claspers held stiff against the body, while gravid females carry the rounded proportions of pregnancy. These changes are not theatrical. They are subtle adjustments in geometry, visible only to observers willing to compare silhouettes over time.
Many coastal shark species rely on estuaries as nursery grounds, where shallow, structured habitat increases juvenile survival by buffering predators and concentrating prey (Heupel et al., 2007). Young sharks enter a world scaled to their size. Warmer water accelerates growth, and complex shoreline geometry provides refuge during early vulnerability.
Some females exhibit fidelity to nursery regions, returning to the same coastal systems that once sheltered them (Heupel et al., 2007). Habitat becomes inheritance. When nursery grounds degrade, the disruption extends beyond a single generation — it interrupts geographic memory embedded in the population itself.

Sandbar sharks (Carcharias plumbeus) invest heavily in durability. They mature late, produce relatively small litters, and rely on long development to generate robust juveniles capable of extended survival (Musick & Ellis, 2005). This strategy favors stability over speed. When mortality rises, recovery unfolds slowly because the species was never designed for rapid turnover.
Sandbar shark reproduction unfolds slowly even by shark standards. Gestation lasts roughly 9–12 months, with litters typically ranging from 6 to 13 pups, though regional variation is common (Musick & Ellis, 2005). Along the mid-Atlantic coast mating generally occurs in spring and early summer, while birthing follows the next year in warmer estuarine margins. The delay is part of the design. Juveniles arrive when prey is abundant and water temperature accelerates growth, aligning birth with a narrow ecological window where survival odds briefly tilt in their favor.
In Onslow County waters, juvenile sandbar sharks use shallow estuary margins as thermal accelerators. Warm, protected water shortens the time required to reach a size less vulnerable to predation. Growth in these early months is not cosmetic; it is survival measured in centimeters. A difference of a few inches can determine whether a young shark passes unnoticed beneath larger predators or becomes part of their diet (Heupel et al., 2007). The nursery functions as a buffer against probability. By compressing early growth into a brief window of ecological generosity, sandbars convert geography into longevity.

Blacktip sharks (Carcharhinus limbatus) align reproduction with seasonal pulses. Birth coincides with warm water and prey abundance, creating a temporary ecological advantage for juveniles. This strategy accepts higher early mortality but compensates through timing — survival synchronized with opportunity (Heupel & Simpfendorfer, 2008).
Blacktip sharks compress their timeline. Gestation averages 10–12 months and litters often contain 1 to 10 pups, with smaller litters more common in northern portions of their range (Heupel & Simpfendorfer, 2008). Mating occurs in late spring and summer; pups are born the following late spring when baitfish concentrations peak in shallow coastal waters. Their strategy hinges on synchronization. Birth is timed not to safety, but to opportunity — a calculated arrival into abundance.
Along our piers in late spring and summer, blacktip juveniles appear in pulses that mirror the prey fields they depend on. Schools of baitfish create moving refuges — density as defense — and young blacktips learn to survive inside motion itself. Survival belongs to individuals able to exploit brief windows, grow fast, and disperse before scarcity returns (Heupel & Simpfendorfer, 2008).

Bonnethead sharks (Sphyrna tiburo) operate on one of the shortest reproductive cycles among coastal sharks. Gestation lasts approximately 4–5 months, and litters commonly range from 4 to 16 pups depending on female size (Hamlett, 2005). Mating generally occurs in late summer, but sperm storage allows fertilization to be delayed until environmental conditions favor gestation. Pups are born in late spring and early summer, entering warm shallow waters that function as immediate nurseries. The speed of the cycle reflects a species built for resilience through repetition — rapid turnover as insurance against instability.
Bonnetheads add evolutionary contingency. Rare cases of parthenogenesis — reproduction without fertilization — demonstrate biological redundancy when mates are scarce (Chapman et al., 2007). Such flexibility underscores a principle of lineage persistence: survival tolerates complexity if complexity improves continuity.
Bonnetheads, often glimpsed in shallow surf or near pilings, compress life history into shorter cycles, allowing populations to respond quickly to environmental change. Unlike many coastal sharks, females are capable of storing viable sperm for extended periods, delaying fertilization until conditions favor successful gestation (Hamlett, 2005). This ability decouples mating from pregnancy, allowing reproduction to align with environmental timing rather than immediate opportunity. Redundancy becomes insurance in a fragmented coastal landscape. Their persistence is not brute strength but flexibility — an evolutionary acknowledgment that coastlines are rarely stable for long (Cortés, 2000).

Sand tiger sharks (Carcharias taurus) represent an uncompromising alternative. Embryos compete within the uterus, and only the strongest survive to birth through intrauterine cannibalism — a process that produces a small number of highly developed juveniles (Hamlett, 2005). From a human perspective the mechanism appears brutal. In evolutionary terms it is a concentrated investment in pre-birth survival.
Sand tiger gestation stretches close to 9–12 months, but the internal competition that defines their development reduces litters to one or two surviving pups per uterus despite a much larger initial embryo count (Hamlett, 2005; Branstetter & Musick, 1994). Mating occurs offshore in cooler months, and births typically follow in spring or early summer. The resulting juveniles are large at birth — already capable hunters — trading quantity for immediate competence. Survival is front-loaded. The species invests in a few individuals built to endure rather than many built to gamble.
For sand tigers occasionally seen near South Topsail Island, this pre-birth selection produces juveniles that enter the water already comparable in size to many adult coastal fish. They arrive as functioning predators. Instead of a long vulnerable childhood, sand tigers begin life past the most dangerous bottleneck. Their subsequent behavior reflects this early security: slow movement, energy conservation, and longevity built on having cleared the lethal threshold before birth (Branstetter & Musick, 1994).
It is tempting to read personality into origin. Yet adult sand tigers move with calm efficiency, rarely engaging in unnecessary conflict. A harsh developmental filter does not predict a harsh adulthood. It simply ensures survival past the most intense threshold.
Together, these strategies map the same coastline through different biological clocks. Some sharks survive by accelerating early growth. Others invest in a few individuals built to last. Still others hedge their future with redundancy. Diversity is not excess — it is resilience expressed through bodies.
Longevity is the silent partner in every reproductive strategy. Long-lived sharks can afford to reproduce slowly, distributing investment across decades. Shorter-lived species compress reproduction into tighter intervals. Neither strategy is superior in isolation. Each is calibrated to environmental tempo (Cortés, 2000).
The coastline holds many clocks at once — tides measured in hours, migrations in seasons, lineage in centuries. Sharks survive by aligning their bodies to the clock that fits their niche. Gestation becomes a wager on stability. Migration becomes inheritance in motion. A nursery becomes infrastructure for continuity.
To observe a pregnant shark offshore is to witness a process already years in motion. The animal carries not only embryos but evolutionary decisions accumulated across millennia: how many to produce, when to move, where to shelter, how long to live. Reproduction is less an event than a continuity. Its future depends not on spectacle, but on whether the slow mathematics of these lives can continue unfolding inside waters still capable of carrying them forward.
Branstetter, S., & Musick, J. A. (1994). Age and growth estimates for the sand tiger in the northwestern Atlantic Ocean. Transactions of the American Fisheries Society, 123(2), 242-254. https://doi.org/10.1577/1548-8659(1994)123<0242:aageft>2.3.co;2
Carlson, A. E., Hoffmayer, E. R., Tribuzio, C. A., & Sulikowski, J. A. (2014). The use of satellite tags to redefine movement patterns of spiny dogfish (Squalus acanthias) along the U.S. East Coast: Implications for fisheries management. PLoS ONE, 9(7), e103384. https://doi.org/10.1371/journal.pone.0103384
Carrier, J. C., Musick, J. A., & Heithaus, M. R. (2012). Biology of sharks and their relatives (2nd ed.). CRC Press.
Chapman, D. D., Shivji, M. S., Louis, E., Sommer, J., Fletcher, H., & Prodöhl, P. A. (2007). Virgin birth in a hammerhead shark. Biology Letters, 3(4), 425-427. https://doi.org/10.1098/rsbl.2007.0189
Cortés, E. (2000). Life history patterns and correlations in Sharks. Reviews in Fisheries Science, 8(4), 299-344. https://doi.org/10.1080/10408340308951115
DeVries, C., Gartland, J., & Latour, R. J. (2025). Patterns in spiny dogfish consumption by sex and maturity stage relate to prey availability and environmental forcing in the Northwest Atlantic. Frontiers in Marine Science, 12. https://doi.org/10.3389/fmars.2025.1621343
Heupel, M., Carlson, J., & Simpfendorfer, C. (2007). Shark nursery areas: Concepts, definition, characterization and assumptions. Marine Ecology Progress Series, 337, 287-297. https://doi.org/10.3354/meps337287
Heupel, M., & Simpfendorfer, C. (2008). Movement and distribution of young bull sharks Carcharhinus leucas in a variable estuarine environment. Aquatic Biology, 1, 277-289. https://doi.org/10.3354/ab00030
Musick, J. A., & Ellis, J. K. (2005). Reproductive evolution of chondrichthyans. In Reproductive Biology and Phylogeny of Chondrichthyes (1st ed., pp. 45-79). Science Publishers.
Pratt, H. L., & Carrier, J. C. (2005). Elasmobranch courtship and mating behavior. In Reproductive Biology and Phylogeny of Chondrichthyes (1st ed., pp. 129-169). Science Publishers.